Pinealon 20mg
$70.00
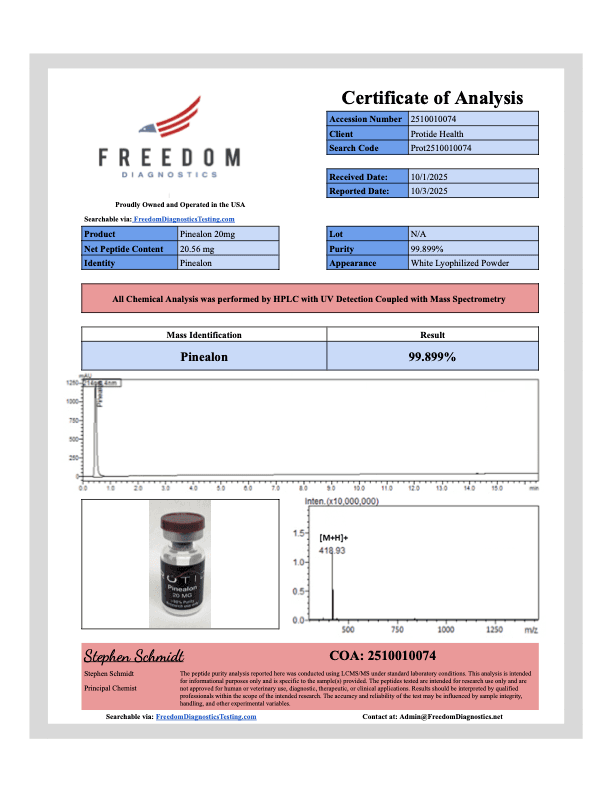
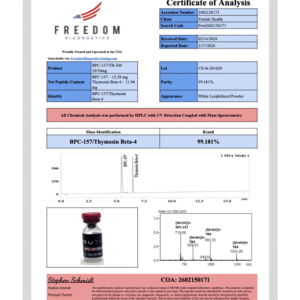
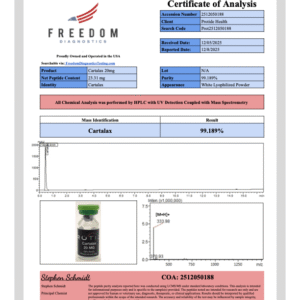
Pinealon peptide studied in preclinical oxidative stress and cellular regulation models. Available in 20mg lyophilized vials. Purity 99% verified by third-party HPLC testing. USA domestic fulfillment. For research use only. Not for human consumption.
Understanding Pinealon in Neuroendocrine and Cellular Research
Pinealon is a synthetic tripeptide (Glu-Asp-Arg) frequently utilized in laboratory settings to examine cellular longevity and genomic interactions. Derived originally from Cortexin (a polypeptide cortex extract), this compound serves as a critical instrument for investigating biological mechanisms. Preclinical models commonly utilize this peptide to explore pathways linked to oxidative stress and neurophysiology.
Exploring the Mechanisms of Pinealon
Laboratory investigations focus on how this peptide modulates specific enzymatic pathways and cellular responses. Researchers study its interactions within strictly controlled environments to gather data on genomic regulation and cellular aging.
Antioxidant and Genomic Pathways
In vitro studies measure the ability of the peptide to alter reactive oxygen species (ROS) in cerebellar granule cells (PubMed). Animal studies examine several specific mechanisms:
-
Downregulation of ROS accumulation in controlled environments.
-
Alterations in ERK 1/2 activation and caspase-3 activity.
-
Measurements of GDF11 and HSPA1A expression during cellular division cycles.
Research Applications in Preclinical Models
The primary role of this tripeptide centers on the regulation of cellular stress responses. Scientists use these compounds in animal models to yield data regarding neuronal viability and neurodegenerative markers.
Oxidative Stress and Hypoxia Studies
Animal studies measure the compound’s influence on biological markers of oxidative damage. Preclinical models observe changes in specific physiological parameters following exposure (PubMed). Research investigates several endpoints:
-
Neuronal necrosis rates in prenatal rat models of hyperhomocysteinemia.
-
Alterations in antioxidant enzyme activity under hypoxic conditions.
-
Cellular survival rates in PC12 cells exposed to hydrogen peroxide.
Cellular Longevity and Cognitive Models
Studies investigate the influence of the peptide on telomere dynamics and spatial learning pathways. Preclinical models measure markers in hippocampal neurons to track biological changes (PubMed). Research examines:
-
Upregulation of irisin expression (linked to telomere elongation) in cellular models.
-
Alterations in the Nr2a/Nr2b ratio during laboratory testing.
-
Amyloid fibril formation rates in Alzheimer’s disease models.
Evaluating Peptide Characteristics
Animal studies evaluate the pharmacokinetic profile of the tripeptide. Preclinical models observe rapid membrane penetration due to its small molecular weight (approximately 418.4 Da), allowing for non-receptor-mediated intracellular interactions (PubMed).
Concluding Thoughts on Pinealon
Pinealon serves as a vital instrument for investigating oxidative stress and neuronal signaling pathways. Preclinical studies continue to examine its influence on genomic regulation, irisin expression, and cellular survival.
References
-
Authors. “Pinealon Cell Viability.” PMC. PubMed
-
Authors. “Pinealon ROS Suppression.” PMC. PubMed
-
Authors. “Pinealon Cell Cycle.” PMC. PubMed
-
Authors. “Pinealon Pharmacokinetics.” PMC. PubMed
Legal Disclaimer
The information provided in this description is for research purposes only. The Pinealon peptide is not approved by the U.S. Food and Drug Administration (FDA) or any regulatory authority for human consumption or therapeutic use. It is intended solely for investigational use in controlled laboratory settings by qualified researchers. Protide Health does not endorse or promote the use of Pinealon peptide in humans or animals outside of approved research protocols. Researchers must comply with all applicable local, state, and federal regulations, including obtaining necessary approvals for experimental use. Consult with regulatory authorities before initiating any research involving Pinealon peptide.
Products sold by Protide Health are for laboratory research purposes only and are not intended for human consumption, medical use, or veterinary use.
Be the first to review “Pinealon 20mg” Cancel reply
Related products
Cellular Longevity Research
Gastrointestinal Research
Best Selling
Cellular Longevity Research
Best Selling
Cellular Longevity Research
Copper Peptides
Best Selling















Reviews
There are no reviews yet.